Gas content of fluid handled
The fluid handled is very rarely a pure liquid (see also Gas separation). Both solid and gaseous substances are usually present in the fluid (see Two-phase flow). Gases can be present in a liquid in dissolved and undissolved form. In the dissolved form, the gas molecules are distributed throughout the liquid and attached to the liquid molecules as a result of physical forces; undissolved gas is present in a liquid in the form of bubbles.
In most cases, the amount of a particular type of dissolved gas cannot exceed the specific limit defined by the properties and condition of the liquid (excluding unstable supersaturated conditions), however, there is practically no upper limit to the amount of undissolved gas a liquid may contain. This is largely dependent on the nature of the fluid's motion. If the fluid is at rest, degassing usually commences rapidly (e. g. the bubbles rise to the surface of the liquid), and the dissolved gas content approaches its maximum value.
A measuring device invented by van Sylke is used to determine the gas content in a liquid. It separates the dissolved gas under vacuum and measures its volume.
A measuring device invented by van Sylke is used to determine the gas content in a liquid. It separates the dissolved gas under vacuum and measures its volume.
Dissolved gases in fluids have an effect on the suction characteristics of centrifugal pumps in so far as a high gas content results in an increase in the required NPSH and thus leads to earlier instances of cavitation.
See Fig. 1 Gas content of fluid handled
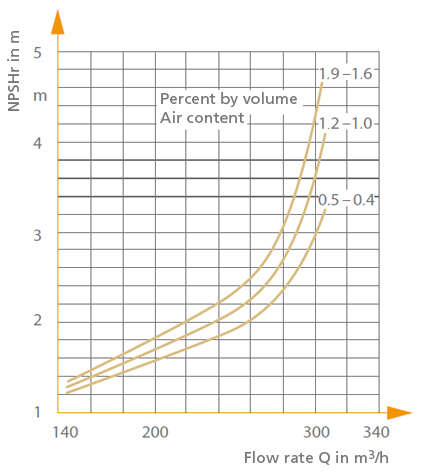
Fig. 1 Gas content of fluid handled: Influence of dissolved air on suction characteristics of a radial centrifugal pump n = 1450 rpm, Qopt = 210 m³/h, Impeller diameter D = 404 mm
Undissolved gas in bubble form influences the overall operating behaviour, and the extent to which gas can be entrained varies widely and continuously according to the design, size and mode of operation of the pump concerned. Fig. 2 Gas content in the fluid handled illustrates the changes in the characteristic curve H(Q), η(Q) of a non-clogging impeller pump (impeller) as a function of the percentage of air entrained.
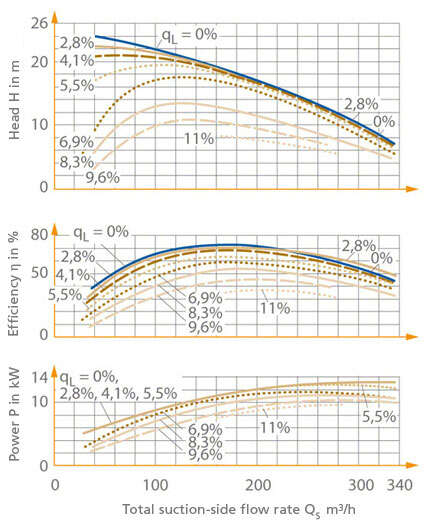
Fig. 2 Gas content of fluid handled: Influence of undissolved air on the operating behaviour of a non-clogging impeller pump handling prefiltered waste water (open three-vane impeller, D = 250 mm, n = 1450 rpm) qL = Air volume/mixture volume (suction nozzle) in %, QS = Suction-side volume flow rate in m³/h
